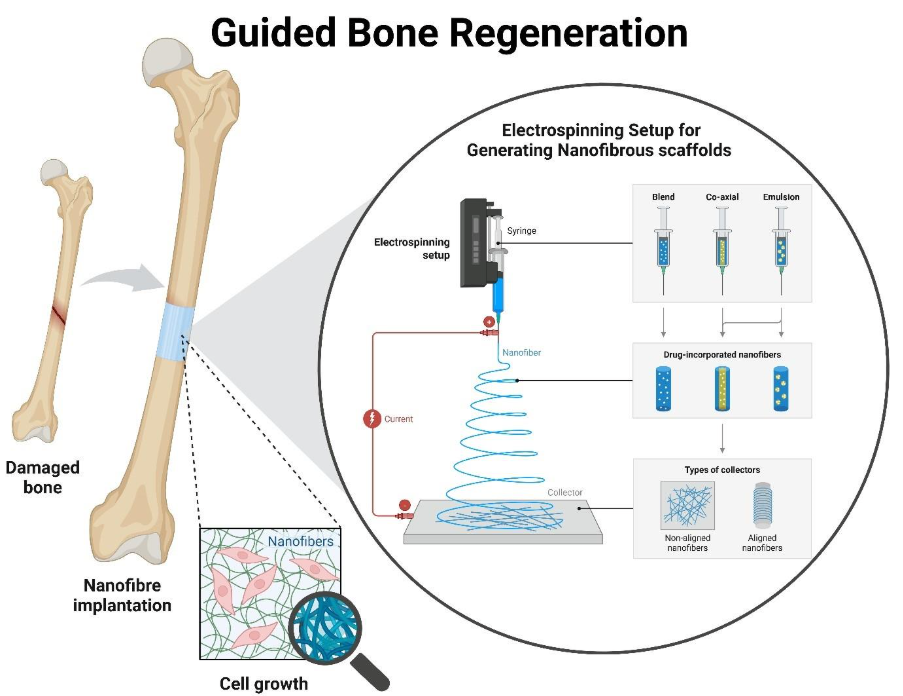
Barrier membranes are central to guided bone regeneration (GBR), yet conventional designs often force trade-offs between cell exclusion, permeability, mechanical stability, and predictable degradation. Electrospun fibrous membranes offer a biomimetic architecture with tuneable porosity and controllable degradation. However, performance alone is not enough. Successful translation requires reproducible manufacturing, scalable processes, and validated structural control. When engineered with consistency in mind, electrospun sheet technologies provide a robust foundation for next-generation GBR devices.
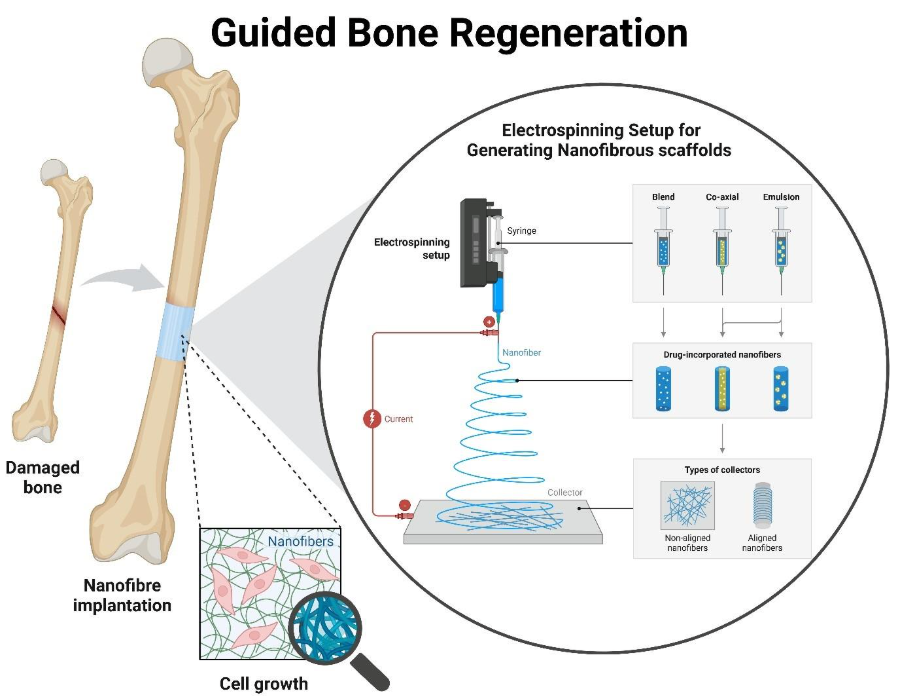
Why barrier design matters in bone regeneration?
Successful bone healing often depends on more than simply filling a defect. In many orthopaedic and dental procedures, the surrounding biological environment actively competes with the regenerative process. Soft tissue can migrate rapidly into the defect site, occupying space intended for new bone formation and compromising long-term outcomes.
GBR addresses this challenge through the use of barrier membranes. These membranes create a protected space that allows osteogenic cells to repopulate the defect while preventing infiltration by faster-growing soft tissue. GBR is widely used in dental implantology, trauma repair, spinal fusion, and the treatment of complex orthopaedic defects.
However, designing a membrane that performs reliably in the clinical environment remains a significant engineering challenge.
The limitations of conventional GBR membranes
Barrier membranes must meet several competing requirements:
- Effective cell exclusion to prevent soft tissue ingrowth
- Sufficient permeability to allow nutrient and fluid transport
- Mechanical stability to maintain space during early healing
- Handling and fixation properties suitable for surgery
- Controlled degradation, where applicable, without premature loss of function
Traditional membrane technologies often require trade-offs. Dense polymer films can provide good barrier function but may limit tissue integration or fluid transport. Collagen-based membranes offer biological compatibility but can degrade unpredictably or lack mechanical strength. Reinforced structures improve stability but increase thickness and complexity.
As clinical expectations evolve, there is growing demand for membranes that combine biological performance, structural integrity, and manufacturing consistency.
For development teams, membrane architecture influences validation strategy, sterilisation approach, and mechanical testing requirements. Changing structure late in development can trigger redesign and regulatory impact, making early material decisions disproportionately important.
Electrospinning: enabling biomimetic barrier architectures
Electrospinning offers a fundamentally different approach to GBR membrane design. Instead of solid films, electrospun membranes are composed of interconnected micro- and nanofibres, creating a highly porous structure that more closely resembles native extracellular matrix.
This fibrous architecture provides several key advantages:
- Selective barrier function: By controlling fibre diameter and packing density, electrospun membranes can be engineered to restrict cellular infiltration while maintaining open pathways for fluid exchange.
- High surface area for tissue interaction: The large surface area promotes protein adsorption and supports early cell attachment at the bone interface, helping to stabilise the regenerative environment.
- Tuneable porosity and thickness: Electrospinning enables precise control over membrane thickness and pore structure, allowing optimisation for different defect sizes and anatomical locations.
- Flexibility and conformability: Thin electrospun sheets can adapt closely to irregular defect geometries, improving surgical handling and defect coverage.
The question is not only whether electrospinning offers performance advantages, but whether the architecture can be consistently controlled and scaled under regulatory scrutiny.
Designing for mechanical stability and space maintenance
One of the primary clinical risks in GBR is membrane collapse into the defect site. This can reduce the available space for bone formation and compromise regeneration.
Electrospun membranes can be engineered to address this through:
- Polymer selection to achieve the required modulus and strength
- Layered or gradient structures to balance flexibility and stiffness
- Thickness optimisation to provide structural support without excessive bulk
For larger or load-sensitive defects, membranes may also be combined with supporting graft materials or fixation systems. The ability to tailor mechanical performance makes electrospinning particularly attractive for orthopaedic applications where defect geometry and loading conditions vary widely.
Membrane collapse or instability during early healing can compromise defect volume and regenerative predictability. If insufficient robustness is identified late in development, redesign can significantly delay timelines.
Controlling degradation to match the healing timeline
In many GBR applications, degradable membranes are preferred to avoid secondary removal procedures. However, premature degradation can lead to loss of barrier function before sufficient bone formation has occurred.
Electrospun degradable polymers allow degradation behaviour to be tuned through:
- Polymer chemistry and molecular weight
- Fibre diameter and surface area
- Membrane thickness and density
This level of control supports the development of membranes that maintain structural integrity during the critical early healing phase and gradually resorb as new tissue matures.
For applications requiring longer-term stability, non-degradable/slow degradable electrospun materials can provide durable barrier performance without structural compromise.
Premature degradation may lead to early loss of barrier function, while prolonged persistence may disrupt remodelling. Achieving the correct balance depends on controlled material selection and fibre architecture.
Adding biological functionality
Beyond passive barrier performance, there is increasing interest in membranes that actively support regeneration.
Electrospun structures are well suited to this approach because they can incorporate:
- Natural polymers such as collagen or gelatin to enhance cell interaction
- Bioactive coatings or blended materials to promote osteogenic activity
- Local delivery of growth factors, antibiotics, or anti-inflammatory agents
Hybrid natural-synthetic fibre systems can combine the mechanical reliability of engineered polymers with the biological cues of native matrix components, supporting more predictable healing outcomes.
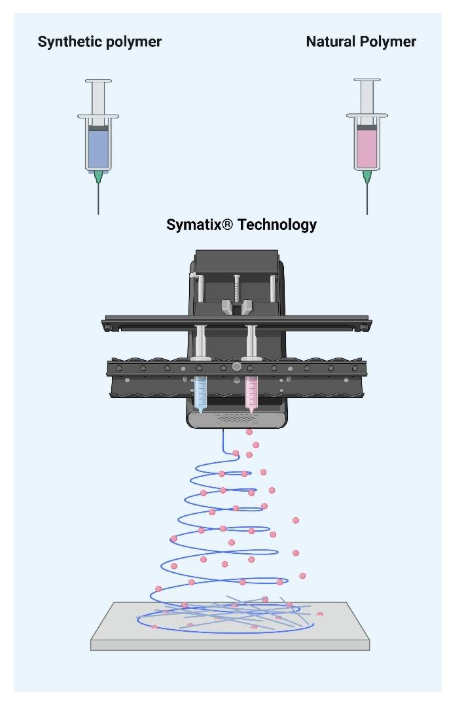
From promising structure to manufacturable product
While electrospinning is widely studied in academic settings, translating fibrous membranes into medical devices requires careful attention to manufacturing considerations. Key factors include:
- Batch-to-batch consistency in fibre morphology and thickness
- Scalable production methods capable of supporting commercial volumes
- Integration into roll-to-roll or sheet-based formats
- Compatibility with sterilisation methods
- Quality control of porosity, mechanical properties, and degradation behaviour
For device developers, reproducibility is decisive. Regulatory submissions require documented control of material architecture and stability through sterilisation and shelf-life. Variability in fibre morphology can translate into variability in mechanical or degradation performance, increasing validation burden. Designing for process control from the outset reduces downstream risk.
A platform approach to GBR membrane design
Electrospun sheet technologies provide flexibility across a range of clinical needs:
- Degradable synthetic systems for resorbable barrier membranes
- Hybrid natural-synthetic structures for enhanced biological interaction
- Non-degradable formats where long-term structural stability is required
- Functional coatings for implant-associated GBR applications
This platform approach allows device developers to tailor material behaviour to specific procedures while maintaining a consistent manufacturing framework.
Looking ahead
As bone regeneration strategies continue to evolve, barrier membranes are expected to play a more active role in guiding and supporting tissue formation. Future designs will likely integrate structural control, biological signalling, and scalable manufacturing into a single solution.
Electrospinning offers a unique combination of architectural precision, material flexibility, and production scalability, positioning fibrous sheet technologies as a strong foundation for next-generation GBR devices across orthopaedic and dental applications.
FAQ?
How do electrospun membranes prevent soft tissue ingrowth while remaining permeable?
By controlling fibre diameter and packing density, pore size can be tuned to restrict cellular infiltration while maintaining nutrient and fluid transport.
Can electrospun GBR membranes maintain space in complex defects?
Mechanical performance depends on polymer selection, thickness, and architecture. Layered or gradient designs can balance flexibility and support, and may be used alongside graft materials where required.
How predictable is degradation in electrospun membranes?
Degradation can be tailored through polymer chemistry and fibre structure. Predictability depends on controlled manufacturing and validation under clinically relevant conditions.
Are electrospun membranes scalable for commercial production?
Commercial translation requires controlled sheet production, fibre consistency, sterilisation compatibility, and validated quality metrics for porosity and mechanical behaviour.