Electrospun Coatings, Membranes and Structures for Cardiovascular Devices
Develop electrospun coatings, membranes, scaffolds and hybrid structures for cardiovascular devices with the evidence, process control and manufacturing pathway needed to assess whether the material decision can survive deployment, validation, scale-up and regulated use.
Book a meeting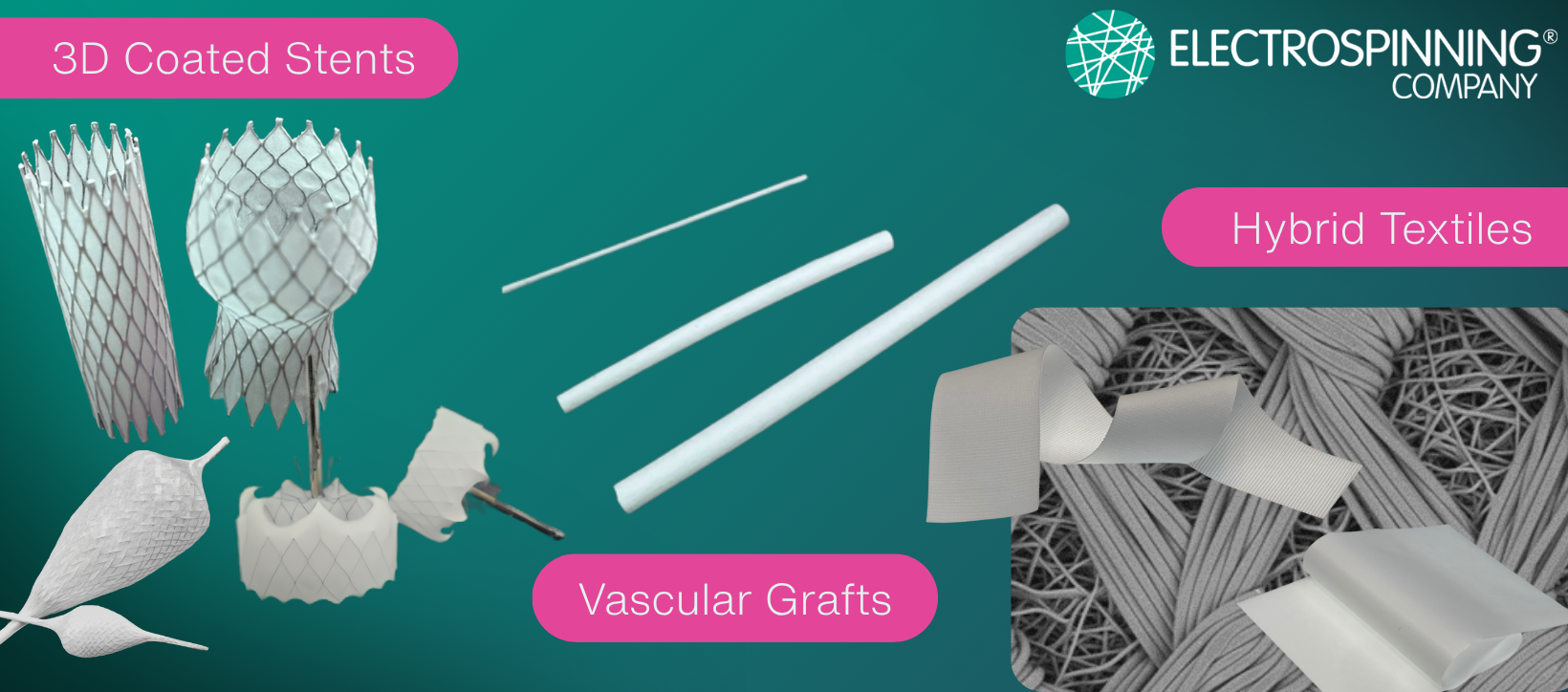
Cardiovascular device teams need material and structure decisions that survive development
The choice of material or coating to deliver robust and sustainable performance in an implantable device is critical to success in cardiovascular surgery.
Electrospinning is relevant in cardiovascular programmes where fibre architecture, coating behaviour, permeability, tissue interaction, deployment integrity or low-profile device integration are performance-critical. Example application contexts include stents, structural heart devices, occlusion devices, vascular grafts, cardiac patches and heart valve structures. Suitability depends on the device architecture, materials, performance requirements and development pathway.
The challenge is not whether an electrospun structure delivers in early testing, but whether it can survive delivery, deployment, fatigue loading, interface demands, and regulatory testing and whether it can be manufactured repeatedly, at appropriate scale and cost.
Applications are not limited to coatings alone. Electrospun fibres can support low-profile surface functionality, 3D nanofibrous patch concepts, tubular scaffold structures, and multilayer architectures that reflect the mechanical behaviour of cardiovascular tissues.
- Relevant to MedTech teams developing minimally invasive and implantable cardiovascular devices
- Particularly suited to programmes evaluating coating, scaffold, membrane or fibre-architecture decisions before validation and regulatory commitment
- Applicable where deployment integrity, permeability, fatigue performance, tissue interaction, low-profile integration or repeatable manufacture are material to device success
- Relevant application contexts may include stents, structural heart devices, occlusion devices, vascular grafts, cardiac patches, heart valve structures
Designed around cardiovascular performance, integration, and manufacturability requirements
Electrospinning can support a range of cardiovascular device goals by linking fibre architecture to the way the device needs to perform, deploy, integrate and scale. Our role is to assess whether electrospinning is the right route, define what must be proven, and develop the structure into a repeatable component, coating, membrane or scaffold suitable for regulated device development.
Controlled coatings on existing devices

Apply electrospun coatings directly onto device surfaces or established device architectures, helping teams improve functionality without redesigning the whole platform.
Precise control of porosity & permeability

Engineer fibre diameter, layer structure and architecture to help manage requirements such as leakage, permeability, tissue response and integration, depending on the application.
Minimally invasive devices compatiblity

Develop fibre structures around the constraints of small, complex or catheter-delivered cardiovascular devices, where flexibility, durability, low-profile design and deployment behaviour may all affect suitability.
Hybrid textile-device structures

Combine electrospun layers with existing textiles, frames or device architectures where the aim is to add controlled surface or structural function without unnecessary platform disruption.
Barrier performance & biological integration

Develop electrospun layers around application-specific barrier, permeability and tissue-interaction requirements, where the device needs both functional separation and biological integration.
From feasibility to decision-grade evidence

Structure feasibility around the evidence needed to decide whether to proceed, refine, pivot or stop before cost, validation and regulatory exposure escalate. Where the route is promising, we help carry the learning forward into development, process engineering, scale-up and regulated manufacturing planning.
In cardiovascular devices, a material that works once is not enough
An electrospun coating, membrane, scaffold or hybrid structure is only useful if it can survive the practical realities of development and use. For cardiovascular teams, that means looking beyond early technical promise to the factors that determine whether the material decision can survive validation, scale-up, regulatory scrutiny and real-world device conditions.
- Deployment survivability — electrospun structures may need to remain stable through delivery, expansion, deployment or handling
- Fatigue performance — cardiovascular applications may expose materials and interfaces to repeated mechanical demands that need to be understood early
- Integrity, adhesion and interface performance — coatings, membranes and hybrid structures need to maintain their intended function in the device environment
- Repeatability under regulated testing — coverage, architecture and performance need to be repeatable, not just promising at concept stage
- Fit with existing device architectures — solutions that build on existing materials, textiles or device frames may reduce development disruption compared with full platform change
A more controlled route to cardiovascular electrospun material development
We help cardiovascular device teams take a more practical step forward by combining electrospinning capability, materials expertise, process development and scalable manufacturing support around real device needs. We can work with established materials and device architectures as well as new concepts, helping teams assess the route from feasibility toward development, validation and commercial device pathways with clearer evidence and less unnecessary disruption.
Caladrix® technology and broader electrospun formats for cardiovascular applications
Caladrix technology provides a low-profile route to electrospun coatings and coverings on medical textiles or device frames. It can be developed around application-specific requirements such as deployment integrity, fatigue performance, surface functionality, permeability and repeatability under regulated testing conditions. Where the device role requires a different structure, we can also develop electrospun membranes, tubular constructs, scaffold architectures or hybrid material formats using the most appropriate platform and process route.
Formats matched to cardiovascular applications
Our platforms support development across a broad range of structural formats, from surface coatings and coverings through to membranes, tubular structures, scaffold architectures and hybrid constructs. The appropriate format is determined by the device role, functional requirements and development pathway, with fibre architecture, geometry and layer composition all configurable to meet the demands of the application. This adaptability means the same core platform can serve projects across a wide range of device categories and therapeutic areas.

Materials selected for application fit
Material options are selected around the application, device pathway and evidence required. Relevant options may include non-degradable polymers such as TPU and PET for mechanically robust coatings, coverings, membranes or scaffold structures, and degradable polyesters such as PCL, PLA and PLGA for resorbable or regenerative applications. Biomolecules and natural polymers may also be considered where the application requires healing, tissue integration or specific biological interaction.

Support from feasibility to scale
By combining proprietary electrospinning technologies, materials expertise, process development, documentation and scalable manufacturing approaches, we help partners move from early feasibility into development, validation, scale-up and commercial device pathways with clearer evidence, stronger process control and reduced programme risk.
As programmes progress, we combine process development, quality controls and data-led manufacturing insight to support repeatability, early issue detection and better decision-making through later-stage development and regulated manufacturing.

Proof beyond feasibility
Our electrospun technologies are not research-stage concepts alone. We have supplied components for five FDA 510(k)-cleared devices on the US market, has more than 10 years of continuous medical device manufacturing experience, and have delivered 100+ feasibility projects across orthopaedics, ophthalmology, soft tissue, cardiovascular and neurology.
We have also supported client products with clinical trial and regulatory submission documentation, and have taken electrospun components beyond feasibility into development, verification and validation, process scale-up and regulated manufacturing.
Explore the material decision before it becomes a programme challenge
We can help cardiovascular device teams assess whether an electrospun coating, membrane, scaffold or hybrid structure is the right fit before the material decision becomes embedded in the programme. The strongest next step is to discuss the application in the context of your device architecture, materials, performance requirements, evidence needs and development stage — then define what must be proven before validation, scale-up and regulatory exposure increase.
Frequently asked questions about electrospinning for cardiovascular devices
Can electrospinning be used on existing cardiovascular devices?
Yes — electrospun coatings can be applied to existing materials or device frames, which can help teams take a lower-risk step without a full platform change.
What kinds of cardiovascular applications are relevant?
Relevant areas include stents, structural heart devices, occlusion devices, vascular grafts, minimally invasive implants, and hybrid textile-device structures.
What materials can be used?
Current source material references TPU, PET, degradable polyesters such as PCL, PLA, and PLGA, plus biomolecules and bioactive components where appropriate to the application.
Why is deployment performance so important?
Because cardiovascular coatings are judged not just on concept performance, but on deployment survivability, integrity, fatigue resistance, and consistency under regulated testing.